Investor Update Issue 02, April 2017
Investor Update: Signs Collaboration Agreement with Zhuhai Blue and O2Vent Successfully Launches in USA
Brisbane, April 4, 2017 AEST (ABN Newswire) - Oventus Medical Ltd ( ASX:OVN) are pleased to provide the Company's latest Investor Update.
ASX:OVN) are pleased to provide the Company's latest Investor Update.
Highlights:
- O2Vent(TM) T launched in the USA and first orders are received
- Oventus strengthens its portfolio offerings with O2Vent(TM) W
- Oventus and Zhuhai Blue Ocean sign strategic collaboration agreement
- Oventus Medical is the lead participant in a successful application for a grant worth $2,950,000 over three years
- Clinical trial indicates O2Vent(TM) T as an alternative to CPAP
- Oventus Manufacturing receives Certificate of Registration certifying the company's Quality Management System complies with the requirements of ISO 13485:20121 and MDSAP Quality Management Certification
1. Message From The CEO
We have achieved a number of significant milestones in the last quarter, expanding the commercialisation efforts of our O2Vent(TM) T device in numerous geographical regions and strengthening our sales and operational systems.
We kick started the year with the launch of O2Vent(TM) T into the US on the back of FDA clearance in September 2016. The US represents a major market for the treatment of obstructive sleep apnoea (OSA). It is estimated that 22 million adults in the US have sleep apnoea with at least 80% of these estimated to be outside of care or not currently being treated effectively with other therapies.
O2Vent(TM) T was officially launched at the G'day USA event in San Francisco on 21st January 2017 celebrating Australian innovation. It was a significant event marking our first product launch into the US market. Since then, records for the first saleable product have been received and appliances have entered manufacturing.
In order to support our launch in the US, Oventus appointed its first US Executive, Mr Steven Wick, as Business Development Manager. Mr Wick is an experienced sales and marketing professional with 26 years of experience across dental and sleep disorder specialties. We believe he is well positioned to lead Oventus Medical into North America and to spearhead the commercialisation of our O2Vent(TM) devices.
Apart from the US, we are also looking to expand into other geographical markets. In February, we signed a landmark agreement with Chinese company Zhuhai Blue Ocean Strategy Medical Ltd (ZHBOS). China represents another massive market opportunity given that the Chinese market is estimated to have over 70 million OSA sufferers, with growing numbers due to the ageing Chinese population.
The collaboration with ZHBOS details terms around clinical trials, training of relevant staff, achievement of relevant regulatory clearances and Chinese marketing preparations. This is a significant step towards penetrating the Chinese market as ZHBOS has over 633 network hospitals, spread throughout China in over 400 cities.
The quarter has also seen a number of operational validation and improvements come through.
Oventus Medical's operating subsidiary, Oventus Manufacturing received a Certificate of Registration certifying that the company's Quality Management System complies with the requirements of The Medical Device Single Audit Program (MDSAP).
MDSAP certification ensures that Oventus' quality management requirements are compliant for multiple countries such as Australia, Europe, USA, Canada, Japan and Brazil. This certification follows the Company's recent ISO 13485 certification in January, which in conjunction, greatly de-risks our recent launch of O2Vent(TM) T into the US. The ISO is a globally recognised certification and a key requirement for the manufacture of medical devices for commercialisation in various geographies.
Most recently, we were also delighted to be the lead participant in a successful grant application worth $2,950,000 of funding spread over three years from the Australian Federal Government for a project to improve sleep apnoea monitoring and treatment.
This project is a key milestone for Oventus in validating its current platform technology and for the expansion of its future product portfolio. We will be working alongside some of Australia's leading researchers such as Neuroscience Research Australia, Western Sydney University, CSIRO and other participants.
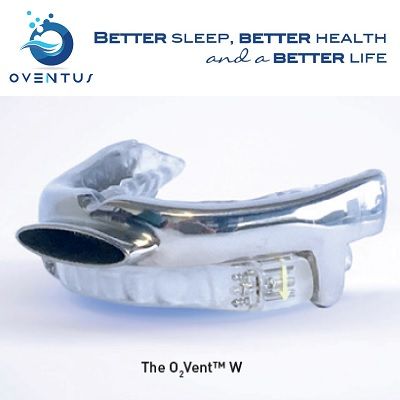
At Oventus we continue to be inspired by the potential of our products and the passion of our team. We have recently launched the O2Vent(TM) W device, a new variation of our O2Vent(TM) T at the American Association of Craniofacial Pain (AACP) Australian Chapter Symposium on Sleep and Pain in Sydney.
This device offers alternative titration capabilities to the O2Vent(TM) T. The wing styled device is the third to join Oventus' range of treatments for OSA. The device utilises a dual mechanism to bring the lower jaw forward, bypassing obstructions of the nose and soft palate.
For the second half of the calendar year, we plan to register additional products with the FDA, gather further clinical evidence to validate the benefit of the unique Oventus airway and to expand on our US launch.
We look forward to keeping you up to date with another exciting quarter ahead.
Thank you for investment and support of the company.
2. Oventus Strengthens Its Portfolio Offerings With O2Vent(TM) W
On World Sleep Day, March 17, Oventus launched its third oral appliance and the latest in its growing portfolio of treatment devices for Obstructive Sleep Apnoea (OSA).
The potentially revolutionary device, O2Vent(TM) W is winged, providing physicians who have long sought a winged device, an option that includes the patented Oventus Airway Technology which acts as a "second nose". Oventus is the only company that has been able to incorporate an airway within the winged device.
The O2Vent(TM) W uses a dual mechanism to stabilise the jaw and move it forward without locking the upper and lower jaw together. The inclusion of the airway is critical as it bypasses the nose and soft palate, where obstructions are major contributors to OSA. With continued research, this has the potential to change the paradigm of care for patients currently suffering from OSA.
3. USA O2Vent(TM) T Launch and First Orders Received
In January 2017, Oventus officially launched O2Vent(TM) T at the G'day USA event in San Francisco.
The event was attended by a representative from the Department of Industry, Innovation and Science, Lindsay Adler the CSIRO General Manager of Global Operations and local clinicians including dentists, sleep clinicians and technology leaders. Records for the first saleable product have been received and appliances have entered manufacturing.
The US launch of the O2Vent(TM) T is a key use for the funds raised from the company's IPO. The US market is a significant market for Oventus with an estimated 22 million adults suffering from OSA. The Oventus team has worked tirelessly six months prior in preparation for the launch which included the submission of the US 510k and FDA clearance back in September 2016.
The Oventus team have laid foundations for a seamless integration with its US subsidiary, Oventus Medical USA based in Delaware. The Company applied for ISO accreditation for the design, development and manufacture of its devices and launched a cloud based enterprise resource planning (ERP) system linking the US subsidiary to the Oventus Medical financial management system.
This ERP system ensures that Oventus' US subsidiary is able to access manufacturing, customer management and patient records which is complaint with the US Health Insurance Portability and Accountability Act - a key requirement for managing patient records in the US.
Finally, the team have set up five pilot sites in various US locations which resulted in the training of clinicians and the successful delivery of evaluation appliances to a small number of patients. To augment its US launch, the team have also partnered with a dental laboratory to receive patient records.The US launch of the O2Vent(TM) T is a key use for the funds raised from the company's IPO. The US market is a significant market for Oventus with an estimated 22 million adults suffering from OSA. The Oventus team has worked tirelessly six months prior in preparation for the launch which included the submission of the US 510k and FDA clearance back in September 2016.
The Oventus team have laid foundations for a seamless integration with its US subsidiary, Oventus Medical USA based in Delaware. The Company applied for ISO accreditation for the design, development and manufacture of its devices and launched a cloud based enterprise resource planning (ERP) system linking the US subsidiary to the Oventus Medical financial management system.
This ERP system ensures that Oventus' US subsidiary is able to access manufacturing, customer management and patient records which is complaint with the US Health Insurance Portability and Accountability Act - a key requirement for managing patient records in the US.
Finally, the team have set up five pilot sites in various US locations which resulted in the training of clinicians and the successful delivery of evaluation appliances to a small number of patients. To augment its US launch, the team have also partnered with a dental laboratory to receive patient records.
4. Clinical Trial Update
Oventus has developed a clinical trial program collaborating with some of Australia's leading research organisations to gather evidence evaluating the O2Vent(TM) T and its unique patented airway.
This follows the completion of the first clinical trial in 2016 which delivered positive results for people with OSA, including those with nasal obstruction, believed to be due to the presence of the airway.
The latest pilot study showed that O2Vent(TM) T reduces the number of respiratory events when used as a standalone device and was effective in the presence of high nasal resistance. The device was also found beneficial to a large percentage of OSA patients who breathe through their mouths and cannot tolerate the Continuous Positive Airway Pressure (CPAP) machine.
Oventus will be gathering more clinical evidence to validate the O2Vent(TM) T as a viable alternative to CPAP.
5. Strategic Collaboration With Chinese Based Zhuhai Blue Ocean
In February, Oventus Medical announced it entered an agreement with Chinese company Zhuhai Blue Ocean Strategy Medical Co., Ltd (ZHBOS).
The collaboration agreement relates to Oventus' proprietary O2Vent(TM) T product, indicated for use during sleep to aid in the treatment of snoring and OSA. The agreement details terms around clinical trials, training of relevant staff, achievement of relevant regulatory clearances and marketing preparations, as they pertain to the Chinese market.
ZHBOS is a Chinese company that has 633 network hospitals, spread throughout China in over 400 cities. Many of the hospitals have ENT sleep clinics some of which recommend Continuous Positive Airway Pressure (CPAP) as the first line of treatment. Oventus' proprietary O2Vent(TM) T device is an innovative alternative to CPAP particularly advantaging patients with nasal obstruction, or those that cannot tolerate, or who refuse CPAP.
6. US Executive Appointed To Support US Launch
During the quarter Oventus appointed its first US Executive Mr Steven Wick, Business Development Manager - North America to support its US launch. Mr Wick is an experienced sales and marketing professional with twenty-six years of experience in the healthcare industry, specifically across the dental and sleep disorder specialties.
Prior to joining Oventus, Mr Wick lead Incisal Edge Sleep, a complete turn-key solution for dentists wishing to add the treatment of obstructive sleep apnoea to their practices. Mr Wick gained a wealth of experience in the dental sleep market in the US over four years as a senior manager of dental sales channels and education for Somnomed before breaking out into his consulting business. During his time at Somnomed, Steven more than doubled his territory's unit sales and this represented one of the most successful markets in the US.
7. CRC-P Successful Grant Application
Oventus Medical is the lead participant in a successful application for a grant in the 2nd round of the CRC-P1 grants that from the Australian Federal Government.
The project will receive $2,950,000 over three years for a project titled, "Targeted therapy for sleep apnoea: A novel personalised approach". The project aims to improve the efficacy, compliance and monitoring of sleep apnoea therapy using a tailored suite of treatments to suit the needs of the individual patient.
To view the complete Investor Update, please visit:
http://abnnewswire.net/lnk/ID4FT3W2
About Oventus Medical Ltd
 Oventus Medical Ltd (ASX:OVN) is a Brisbane based medical device company that is commercialising a unique treatment platform for the treatment of sleep apnoea and snoring. Unlike other oral appliances or CPAP interfaces, the Oventus devices have a unique and patented airway within the treatment platform that allows air to flow to the back of the mouth unobstructed while maintaining an oral seal and stable jaw position, bypassing multiple obstructions from the nose, soft palate and tongue, reducing airway collapsibility and managing mouth breathing while maintain a stable airway with or without nasal CPAP. They are particularly designed for the many people that have nasal obstructions and consequently tend to mainly breathe through their mouth. While it may seem counterintuitive, this technology actually manages mouth breathing by converting it to device breathing and normalising ventilation. The O2Vent(TM) is designed to allow nasal breathing when the nose is unobstructed, but when obstruction is present, breathing is supplemented via the airways in the appliance.
Oventus Medical Ltd (ASX:OVN) is a Brisbane based medical device company that is commercialising a unique treatment platform for the treatment of sleep apnoea and snoring. Unlike other oral appliances or CPAP interfaces, the Oventus devices have a unique and patented airway within the treatment platform that allows air to flow to the back of the mouth unobstructed while maintaining an oral seal and stable jaw position, bypassing multiple obstructions from the nose, soft palate and tongue, reducing airway collapsibility and managing mouth breathing while maintain a stable airway with or without nasal CPAP. They are particularly designed for the many people that have nasal obstructions and consequently tend to mainly breathe through their mouth. While it may seem counterintuitive, this technology actually manages mouth breathing by converting it to device breathing and normalising ventilation. The O2Vent(TM) is designed to allow nasal breathing when the nose is unobstructed, but when obstruction is present, breathing is supplemented via the airways in the appliance.
For more information on Oventus' Sleep Treatment Platform, please visit https://www.oventus.com
| ||
|









